
Chinese scientists have built a nuclear battery that can produce power for up to 50 years without being recharged. The technology, which contains a radioactive isotope, or version of nickel, as its power source, will be the first of its kind available for general purchase, Betavolt company representatives said on January 8, 2024 (statement in Mandarin).
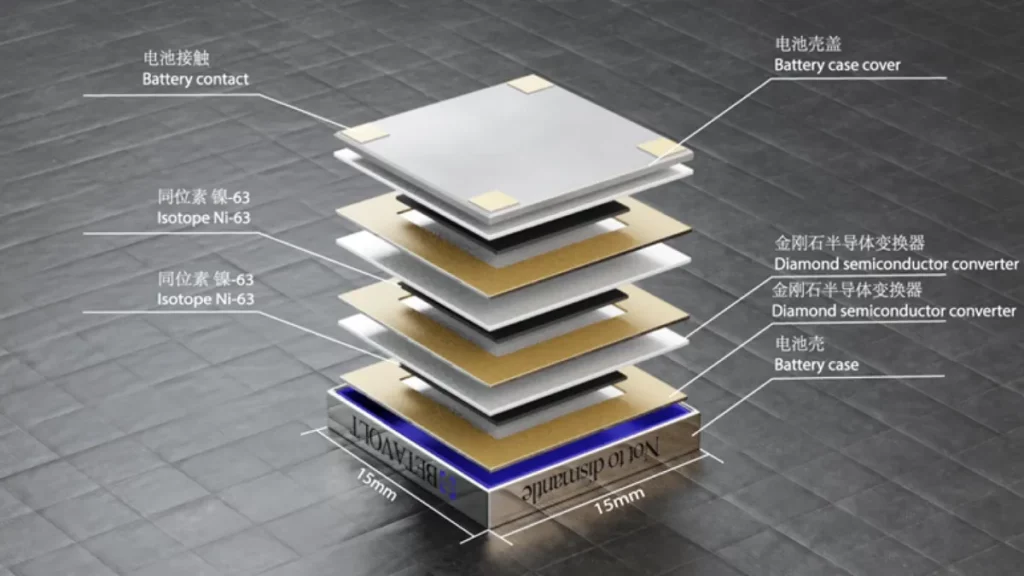
The new battery, dubbed “BV100”, is smaller than a coin, measuring 15 x 15 x 5 millimeters, and generates 100 microwatts of power. If approved for use in devices like smartphones, future generations of the battery would ultimately remove the need to ever charge them, company representatives said.
The challenge of how to access the maximum power from a single battery is still an area of research and Betavolt plans to launch a 1 watt battery in 2025 which is much closer to the 2 – 6 W required by a standard cell phone. In the meantime, the company suggested combining its batteries in parallel to increase the power directed to a device.
The company also plans to research the use of different nuclear isotopes in future versions of its nuclear battery, including strontium-90, promethium-147 and deuterium, which can last between two and 30 years in a device.
But a U.S. materials scientist with a small consultancy group linked to the University of Florida, is skeptical. He claims the size of Betavolt’s device means it contains relatively little radioisotope and it produces just 0.01% of the electricity required. He says the Chinese device is within the range for a pacemaker or perhaps a passive wireless sensor, but that in its current form he doubts it would have enough power to run a cell phone.
Nuclear batteries are a well-established technology. First developed in the early 1950s, these devices harness the energy released when radioactive isotopes decay into other elements. As long as the radioactive element is decaying, the battery will continue generating power. It means nuclear batteries typically have decades-long lifespans and are commonly used to power spacecraft or automated scientific stations — where equipment can be left unattended for years at a time. They’re also used in heart pacemakers.
Betavolt’s battery uses nickel-63 as the radioactive source, which decays into metallic copper-63 through a beta pathway. The copper is stable (non-radioactive), but the nickel-63 isotope has a half life of 101.2 years1. The radioactive decay emits electrons, which can be used as a source of electricity.
The BV100 uses a semiconductor layer to capture these electrons and conduct them through the battery in an organized fashion. A semiconductor is in-between a conductor like a metal and an insulator like rubber. The electrons can move if they have sufficient energy and these can be control they moved.
Betavolt’s battery sandwiches the radioactive nickel between two ultrathin plates of diamond, a particularly efficient semiconducting material, converting the electrons released by radioactive decay into a usable electric current.
These radioisotopes pose little danger if they’re used in space, for example, but need shielding with materials that can absorb harmful radiation if used in devices like pacemakers or in future smartphones to be considered safe.
The radiation needs to be shielded from the body. Radiation protection, often made of materials such as lead or tungsten, is usually integrated into the battery design, and that can be impractical for small scale devices. However, despite the need for weighty shielding, nuclear batteries do boast a much higher energy density than conventional lithium-ion batteries — ten times as much according to Betavolt.
The challenge of how to access the maximum power from a single battery is still an area of research and Betavolt plans to launch a 1 watt battery in 2025 which is much closer to the 2 – 6 W required by a standard cell phone. In the meantime, the company suggested combining its batteries in parallel to increase the power directed to a device.
The company also plans to research the use of different nuclear isotopes in future versions of its nuclear battery, including strontium-90, promethium-147 and deuterium, which can last between two and 30 years in a device.
- Half-life, in radioactivity, is the interval of time required for one-half of the atomic nuclei of a radioactive sample to decay (change spontaneously into other nuclear species by emitting particles and energy). ↩︎

Source: Live Science (edited version), January 25, 2024. https://www.livescience.com/technology/engineering/betavolt-bv100-radioactive-battery-can-last-50-years-coming-in-2025
See review of Betavoltaic technology by Chinese scientists:
‘Review—Betavoltaic Cell: The Past, Present, and Future’, Chunlin Zhou et al 2021 ECS J. Solid State Sci. Technol., https://doi.org/10.1149/2162-8777/abe423